Non-porous reference electrodes in s::can pH and ORP sensors offer a technical advantage over all other pH and ORP sensors with a junction between the sample and the electrolyte.
How does it work?
Traditional pH sensors have a junction (or two) between the sample and the reference electrode, this enables electrical contact between the sample and the reference electrode which is fundamental to pH measurement. The sample causes the contamination of the electrolyte surrounding the Ag/AgCl reference electrode (usually 3M KCl) over time, this is the reason that pH sensors have a tendency to drift and eventually fail.
A non-porous polymer coating on a sensor provides an electrochemical connection between the sample and the reference electrode without a junction. This means that the electrolyte does not get contaminated or poisoned over time, nor does physical fouling of the sensor make any difference to accuracy. As long as any coating on the sensor is conductive and not excessively thick, pH measurement accuracy will not be affected.
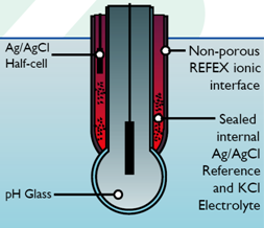
What does this mean in practice?
- Less calibration and maintenance
In raw surface water, the sample can be cold and have low ionic strength. In a traditional pH sensor, the electrolyte quickly diffuses out the sensor through the junction because of the concentration gradient causing the sensor to drift and require recalibrating. Because the non-porous electrode doesn’t lose its electrolyte, it doesn’t drift and maintenance is reduced as the electrode does not need its electrolyte replaced. Less drift, less calibration and less maintenance.
- Lower operational expenditure
Fats, oils and greases, insoluble manganese deposits, proteins etc all can clog or coat a traditional pH sensor with a junction. Without the junction, a non-porous electrode can continue to perform even when coated. Fewer electrode replacements required means lower operational expenditure.
- Faster response time
A single-junction pH sensor requires the diffusion of the ions in the sample through the junction to react to changes. Using a double junction electrode extends the life of the sensor, but slows the response time. None of this is a problem with a non-porous electrode. The rapid response to pH changes in a non-porous electrode means no over-shooting when it comes to chemical dosing.
How can I find out more?
